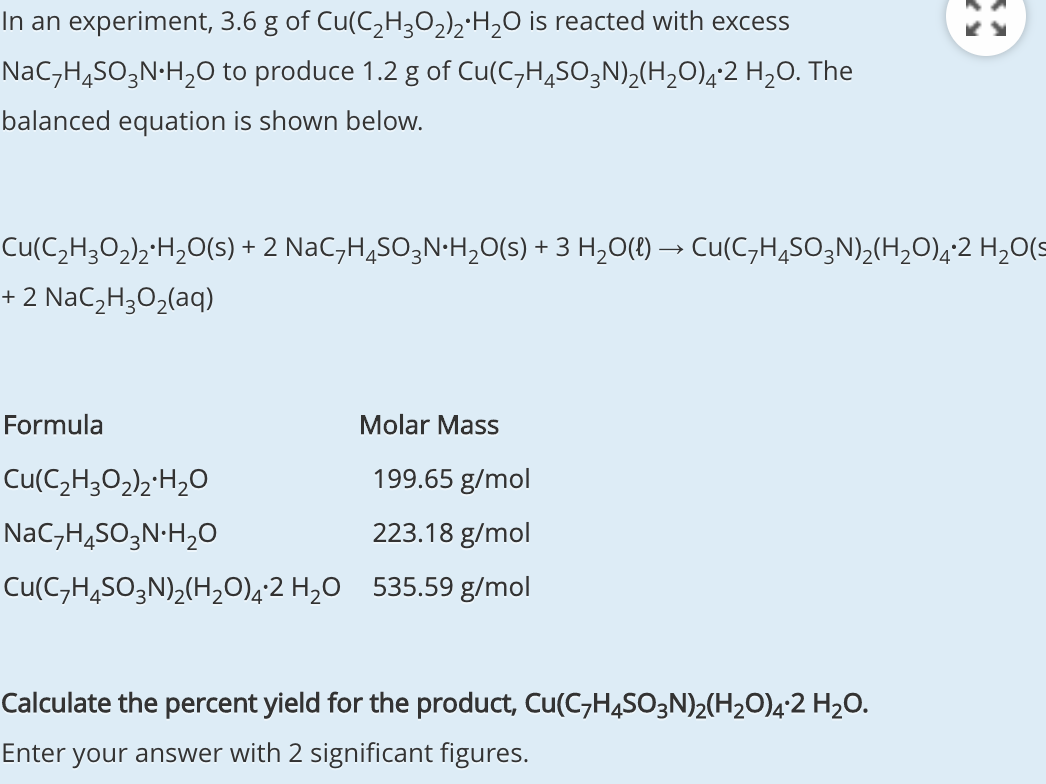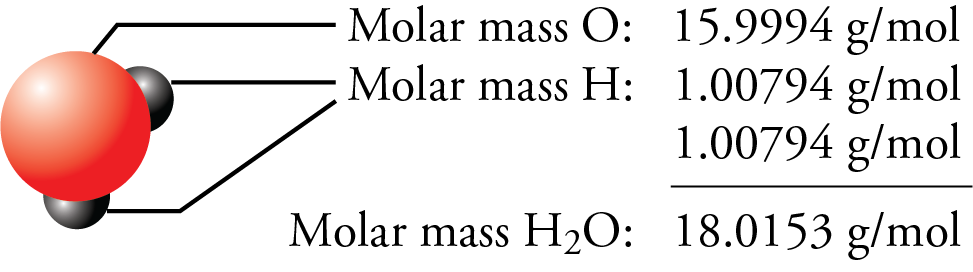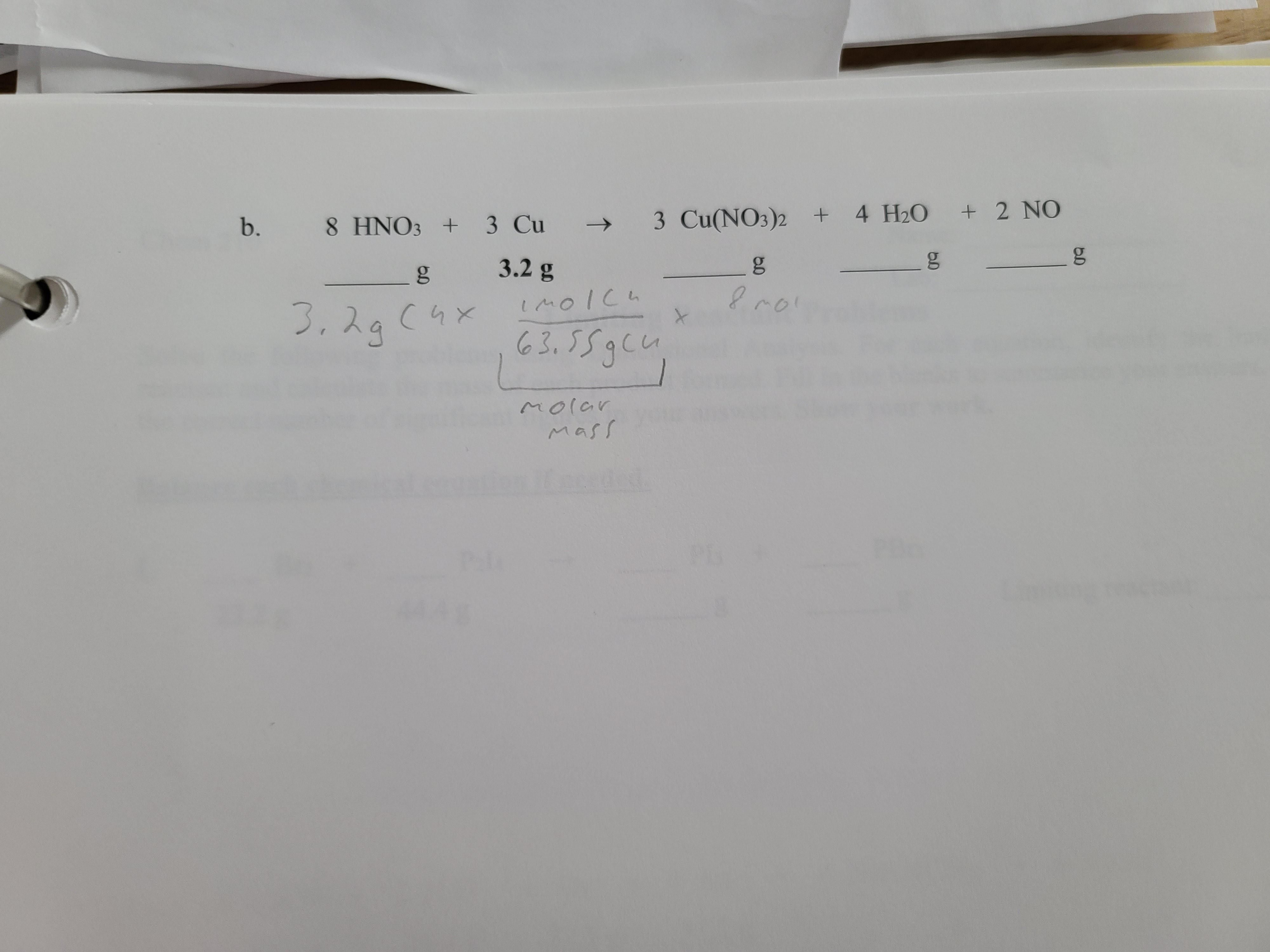Molar mass of co(NO3)2 6H2O is 290grams given mass is 30grams in 4 3litres of solution calculate molarity - Chemistry - - 13950035 | Meritnation.com

Calculate the molar mass of he following : (i) H2O (ii) CO2 (iii) CH4 Ncert solutions for Class 11th - Brainly.in

The molar mass of H2O is 18.01 g/mol. The molar mass of O2 is 32.00 g/mol. What mass of H2O, ins - Brainly.com

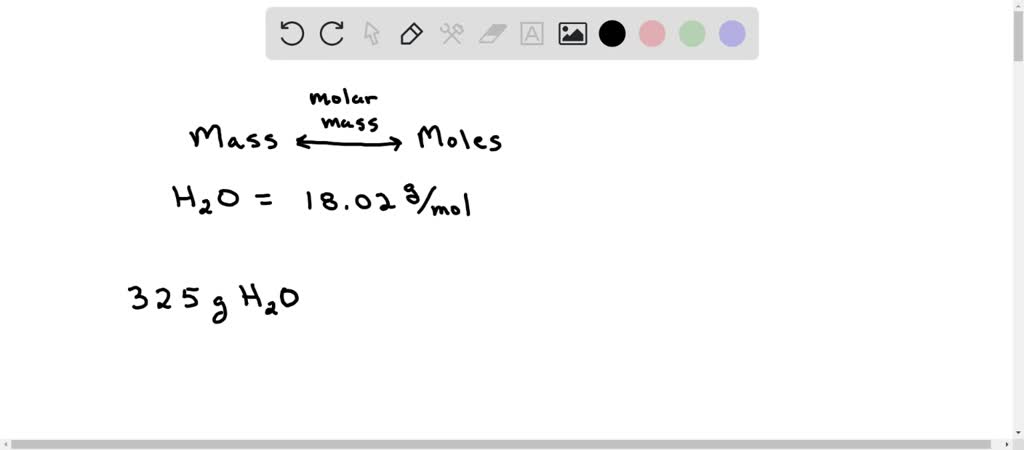
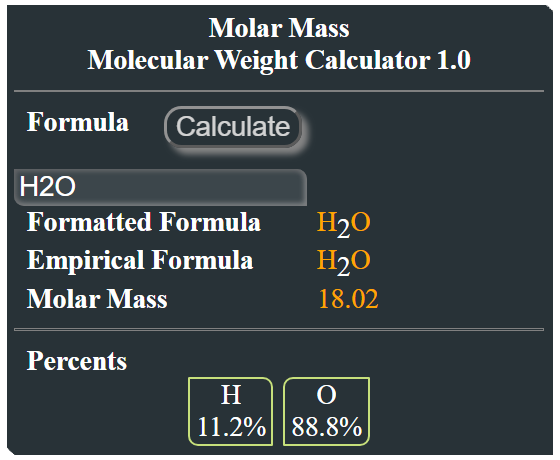
Calculate Calculate the molar mass of H2O - Chemistry - Some Basic Concepts of Chemistry - 12618097 | Meritnation.com

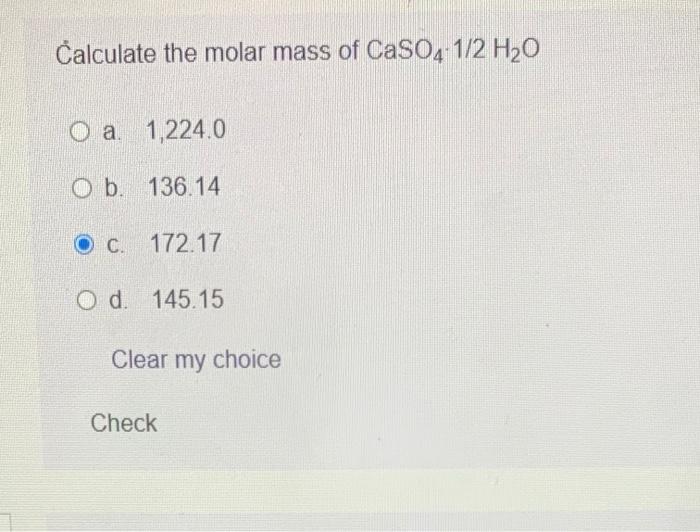
SOLVED: What mass in grams of the dihydrate H2C2O4 • 2 H2O (molar mass = 126.0 g/mol) is needed in order to deliver 16.4 grams of H2C2O4 (molar mass = 90.0) to