Single vs double atom catalyst for N2 activation in nitrogen reduction reaction: A DFT perspective - Qian - 2020 - EcoMat - Wiley Online Library

New insight into N2 adsorption and ion-exchange features of CuMFI with different Si/Al ratios - New Journal of Chemistry (RSC Publishing)

Azide ion ( N3^ - ) exhibits on N - N bond order of 2 and may be represented by resonance structures, I, II and III given below. Select correct statements.
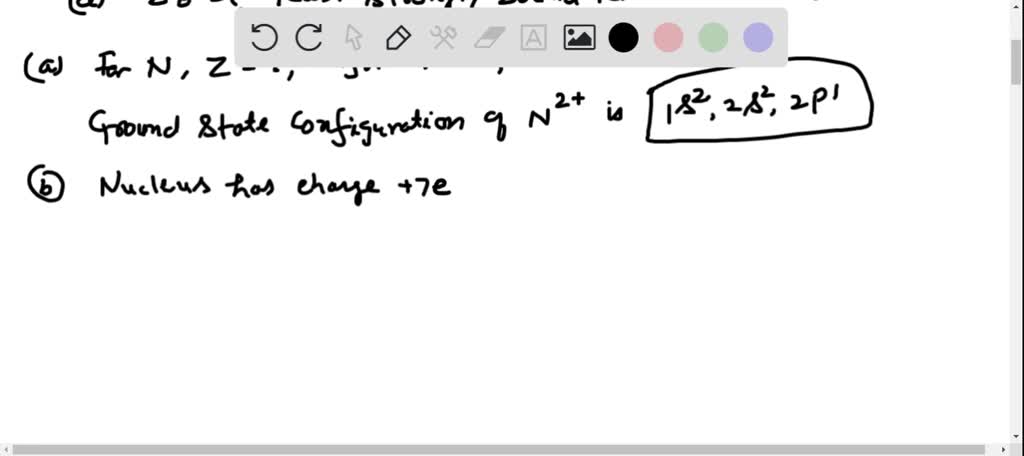
SOLVED:(a) The doubly charged ion N^2+ is formed by removing two electrons from a nitrogen atom. What is the ground-state electron configuration for the N^2+ ion? (b) Estimate the energy of the

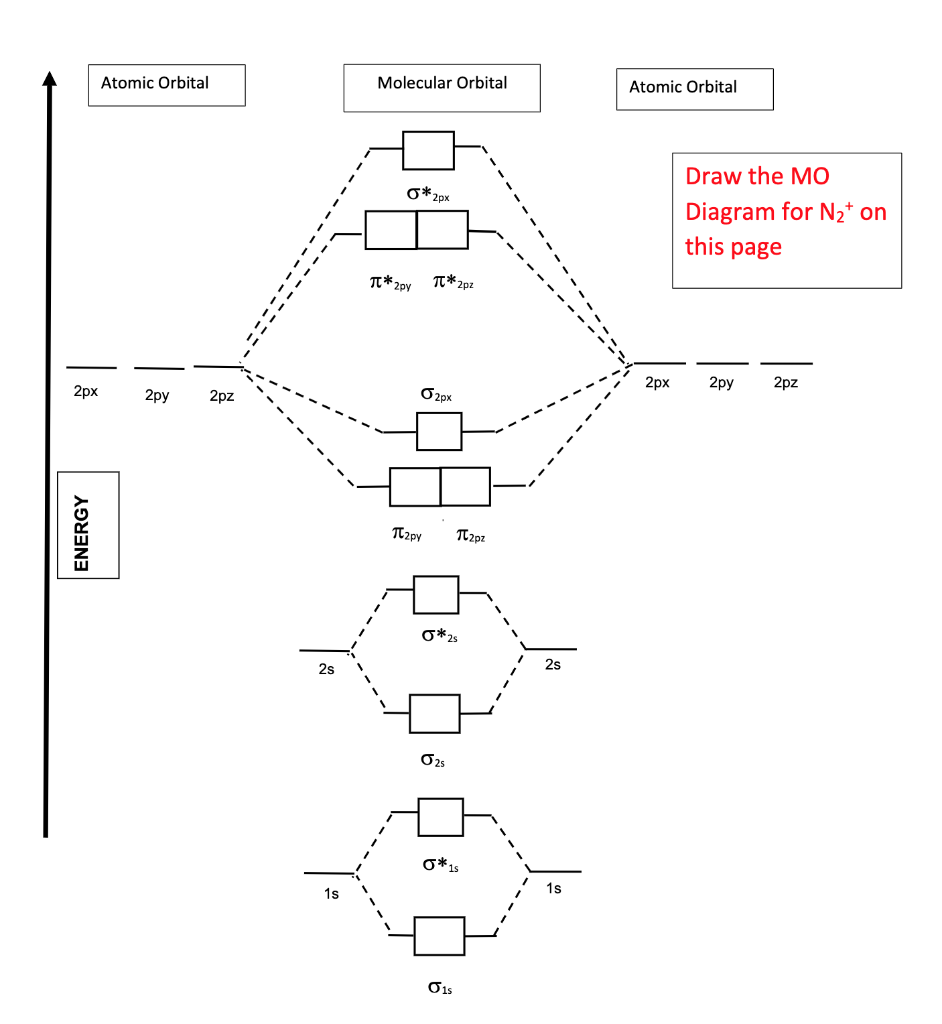
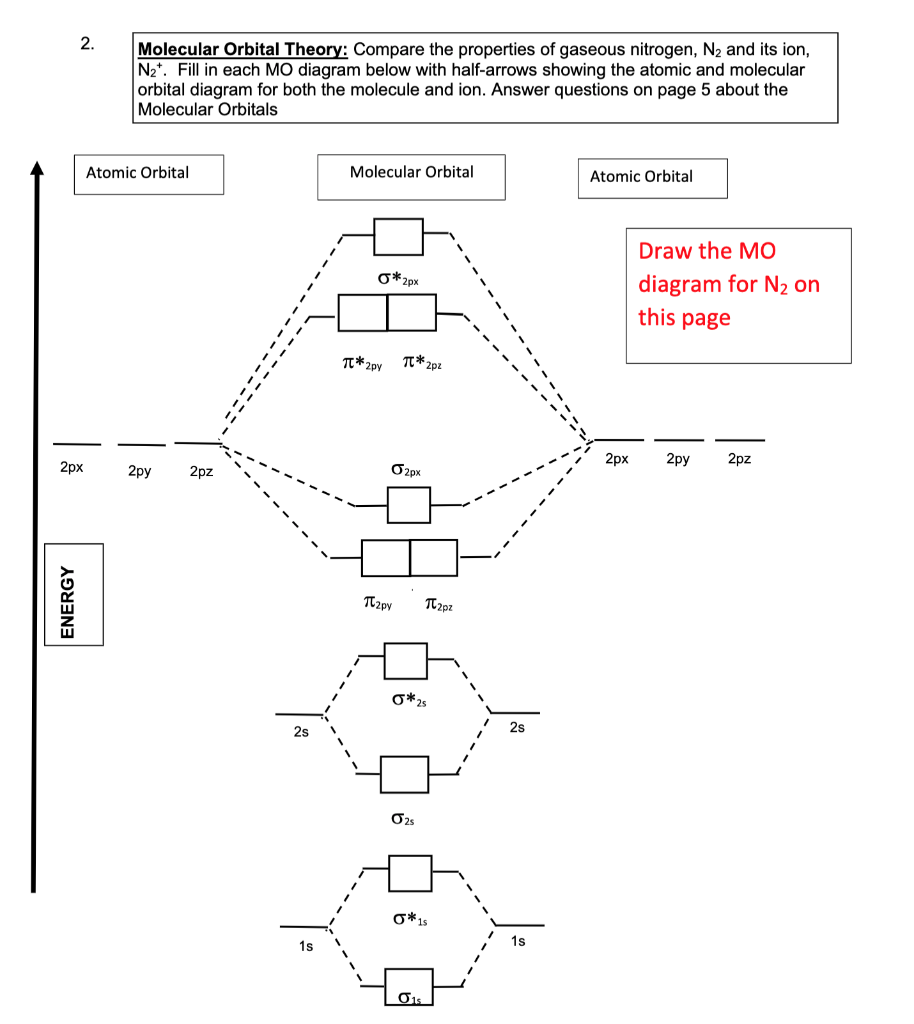
In the molecular orbital diagram for the molecular ion, N^ + 2 , the number of electrons in the σ2p molecular orbital is :

Potential-energy diagram of molecular nitrogen and its ion. The ionic... | Download Scientific Diagram