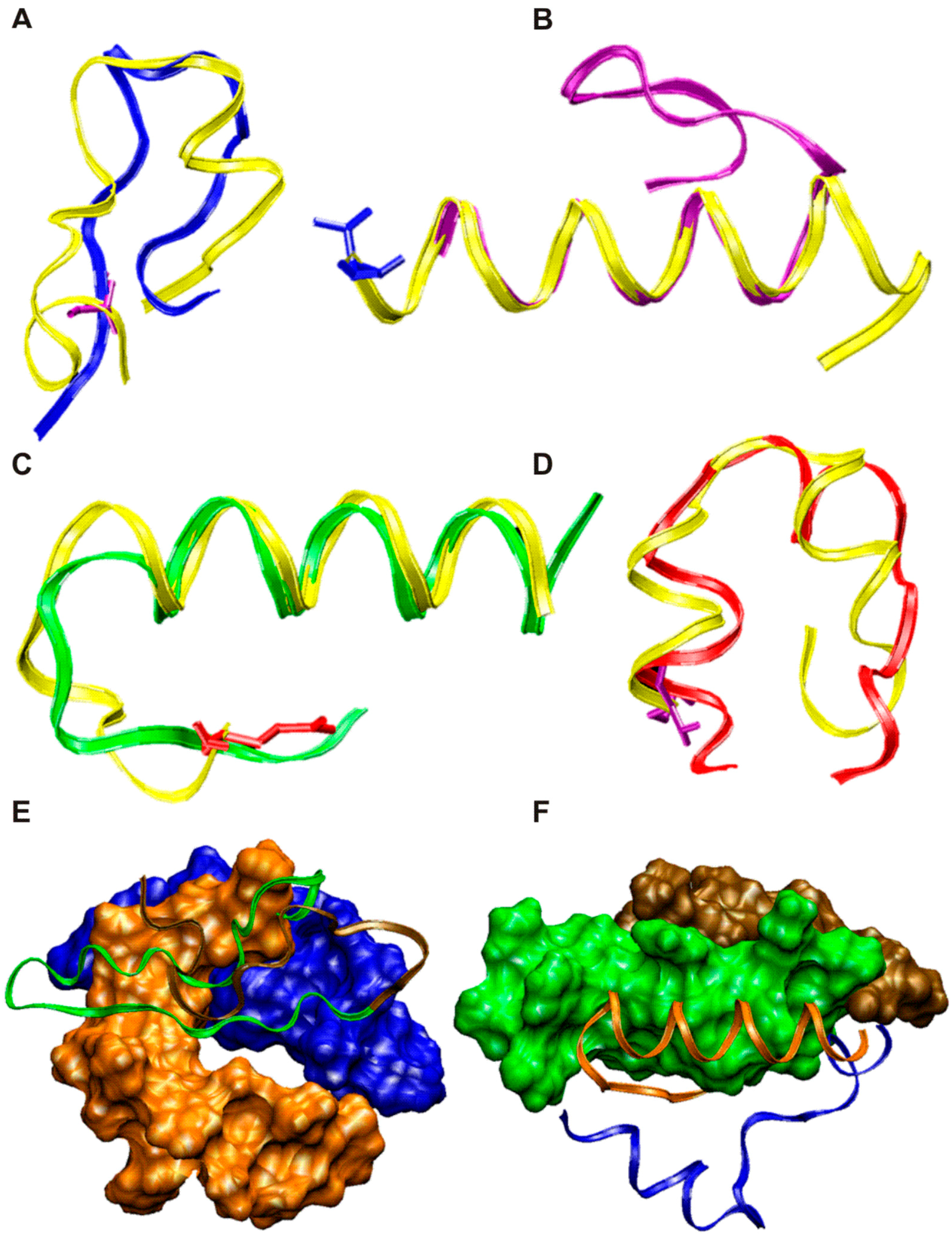
N-Methylated Cyclic RGD Peptides as Highly Active and Selective αVβ3 Integrin Antagonists | Journal of Medicinal Chemistry

Development of a solid‐phase traceless‐Ugi multicomponent reaction for backbone anchoring and cyclic peptide synthesis - Jobin - 2019 - Peptide Science - Wiley Online Library

PDF) The synergy of ChemMatrix resin® and pseudoproline building blocks renders Rantes, a complex aggregated chemokine | Judit Tulla-puche - Academia.edu

An Activated O → N Acyl Transfer Auxiliary: Efficient Amide-Backbone Substitution of Hindered “Difficult” Peptides | The Journal of Organic Chemistry
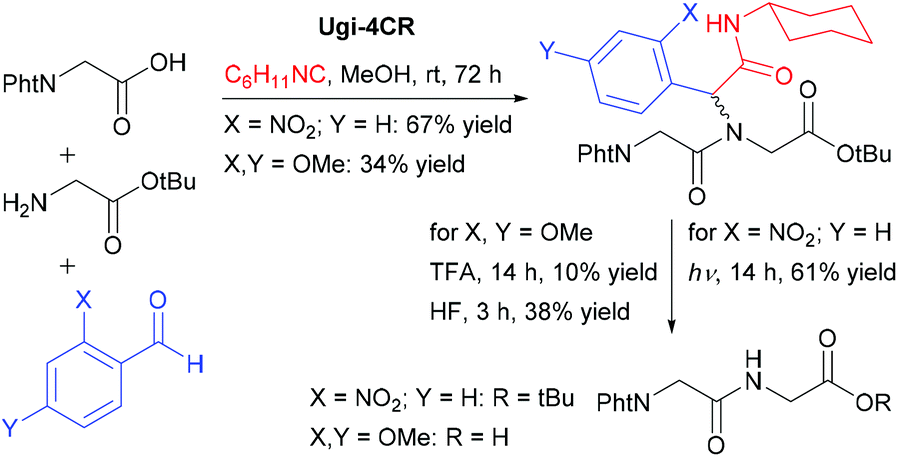
Toward solid-phase peptide fragment ligation by a traceless-Ugi multicomponent reaction approach - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C6OB02342H